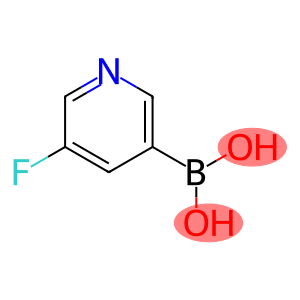
5-fluoro-3-pyridylboronic acid
5-Fluoropyridin-3-ylboronic acid
CAS: 872041-86-6
Molecular Formula: C5H5BFNO2
5-fluoro-3-pyridylboronic acid - Names and Identifiers
5-fluoro-3-pyridylboronic acid - Physico-chemical Properties
| Molecular Formula | C5H5BFNO2 |
| Molar Mass | 140.91 |
| Density | 1.34±0.1 g/cm3(Predicted) |
| Melting Point | 233-236 |
| Boling Point | 292.9±50.0 °C(Predicted) |
| Flash Point | 130.9°C |
| Solubility | Soluble in hot methanol. |
| Vapor Presure | 0.000813mmHg at 25°C |
| Appearance | Crystalline Powder |
| Color | White to off-white |
| pKa | 5.85±0.10(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | 1.507 |
| MDL | MFCD07368243 |
5-fluoro-3-pyridylboronic acid - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | R37/38 - Irritating to respiratory system and skin. R41 - Risk of serious damage to eyes R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S39 - Wear eye / face protection. S37 - Wear suitable gloves. |
| HS Code | 29319090 |
| Hazard Note | Irritant/Keep Cold |
5-fluoro-3-pyridylboronic acid - Introduction
Acid, also known as acid, is an organic compound. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: acid is a colorless to off-white crystalline or powdery solid.
-Solubility: Soluble in common organic solvents such as ethanol, methanol and dichloromethane.
Use:
-Chemical reagent: acid is commonly used as a reagent in organic synthesis to construct carbon-carbon bonds and carbon-boron bonds. It can participate in the reaction of phenyl and pyridine.
-Drug synthesis: This compound has an important role in the field of drug synthesis and can be used to synthesize anti-cancer drugs, antiviral drugs and other biologically active molecules.
Preparation Method:
-Cr acid can be synthesized by a variety of methods, commonly used methods include:
1. under alkaline conditions, 5-fluorine -3-pyridine formaldehyde and three phenyl boron reaction, acid hydrolysis after the product.
2. the 5-fluorine -3-pyridine ketone and sodium triphenyl borate reaction, the product after acid hydrolysis.
3. can also be through other methods, such as C- C bond and C- B bond hydrogenation reduction reaction.
Safety Information:
-the safety of acid is high, but it is still necessary to follow routine laboratory operations and personal protective measures.
-In case of fire, suitable fire extinguishing equipment shall be used to extinguish the fire.
-Avoid inhaling, chewing or eating the compound when used.
-To be stored in a closed container, away from fire sources, heat sources and oxidants.
-Detailed safety operation guidelines and emergency handling procedures should follow relevant safety documents and laboratory regulations.
Nature:
-Appearance: acid is a colorless to off-white crystalline or powdery solid.
-Solubility: Soluble in common organic solvents such as ethanol, methanol and dichloromethane.
Use:
-Chemical reagent: acid is commonly used as a reagent in organic synthesis to construct carbon-carbon bonds and carbon-boron bonds. It can participate in the reaction of phenyl and pyridine.
-Drug synthesis: This compound has an important role in the field of drug synthesis and can be used to synthesize anti-cancer drugs, antiviral drugs and other biologically active molecules.
Preparation Method:
-Cr acid can be synthesized by a variety of methods, commonly used methods include:
1. under alkaline conditions, 5-fluorine -3-pyridine formaldehyde and three phenyl boron reaction, acid hydrolysis after the product.
2. the 5-fluorine -3-pyridine ketone and sodium triphenyl borate reaction, the product after acid hydrolysis.
3. can also be through other methods, such as C- C bond and C- B bond hydrogenation reduction reaction.
Safety Information:
-the safety of acid is high, but it is still necessary to follow routine laboratory operations and personal protective measures.
-In case of fire, suitable fire extinguishing equipment shall be used to extinguish the fire.
-Avoid inhaling, chewing or eating the compound when used.
-To be stored in a closed container, away from fire sources, heat sources and oxidants.
-Detailed safety operation guidelines and emergency handling procedures should follow relevant safety documents and laboratory regulations.
Last Update:2024-04-09 02:00:48
Supplier List
Spot supply
Product Name: 5-Fluoropyridine-3-boronic acid Visit Supplier Webpage Request for quotationCAS: 872041-86-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-Fluoropyridin-3-ylboronic acid Request for quotation
CAS: 872041-86-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 872041-86-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 5-Fluoropyridin-3-ylboronic acid Request for quotation
CAS: 872041-86-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 872041-86-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 5-Fluoropyridin-3-ylboronic acid Request for quotation
CAS: 872041-86-6
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 872041-86-6
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Spot supply
Product Name: 5-Fluoropyridine-3-Boronic Acid Visit Supplier Webpage Request for quotationCAS: 872041-86-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 5-Fluoropyridine-3-boronic acid Visit Supplier Webpage Request for quotationCAS: 872041-86-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-Fluoropyridin-3-ylboronic acid Request for quotation
CAS: 872041-86-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 872041-86-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 5-Fluoropyridin-3-ylboronic acid Request for quotation
CAS: 872041-86-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 872041-86-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 5-Fluoropyridin-3-ylboronic acid Request for quotation
CAS: 872041-86-6
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 872041-86-6
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Spot supply
Product Name: 5-Fluoropyridine-3-Boronic Acid Visit Supplier Webpage Request for quotationCAS: 872041-86-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




